In the rapidly advancing field of cardiovascular therapeutics, certain patents stand out for their clinical relevance and scientific depth. EP3536712B1 is one of them.
This European patent, held by Regeneron Pharmaceuticals Inc., covers methods for reducing Lipoprotein(a) levels through the administration of PCSK9 inhibitors. The innovation is particularly important for treating patients at elevated risk of cardiovascular disease. The patent is currently under appeal, with Regeneron challenging Amgen across multiple jurisdictions.
Rather than focusing on the legal dimensions of the case, this article uses the Global Patent Search (GPS) platform to explore patents with similar therapeutic strategies. We’ll examine technologies that target PCSK9, influence Lp(a) levels, or deploy comparable biological mechanisms.
For IP analysts, life sciences professionals, or anyone monitoring trends in cardiovascular treatments, this is a data-driven look at how related innovations cluster around EP3536712B1.
Understanding Patent EP3536712B1
EP3536712B1 discloses methods for lowering serum lipoprotein(a) [Lp(a)] levels in patients. The invention involves administering a PCSK9 inhibitor, specifically an antibody or antigen-binding fragment that binds PCSK9. These inhibitors block PCSK9’s function, resulting in reduced Lp(a) levels in patients with elevated cardiovascular risk. The composition is designed for individuals who are not on statin therapy at the time of treatment.
Its Four Key Features Are
#1. PCSK9 inhibition: Use of an antibody or fragment that specifically binds and inhibits human PCSK9.
#2. Target patient group: Individuals with serum Lp(a) levels above 30 mg/dL and elevated cardiovascular or thrombotic risk.
#3. Non-statin context: Treatment is administered to patients not currently on therapeutic statin regimens.
#4. Disease applications: Applicable to patients at risk of coronary artery disease, stroke, pulmonary embolism, or Familial Hypercholesterolemia. This aligns with the work of healthcare companies advancing therapeutic design across lipid metabolism and cardiovascular health.
This patent presents a novel therapeutic approach to lowering Lp(a), a persistent and under-treated cardiovascular risk factor. It highlights the expanding clinical utility of PCSK9 inhibitors beyond LDL cholesterol management.
Related Read: See how patents like US12245996B2 ensure norepinephrine remains stable under stress conditions, resonating with patents like EP3536712B1 that focus on thermal resilience in injectable drugs.
Similar Patents As EP3536712B1
To explore the innovation landscape surrounding EP3536712B1, we ran the patent through the Global Patent Search tool. Below is a quick glimpse of the GPS tool in action:

Source: Global Patent Search
This analysis surfaced a list of related patents that share technical similarities in PCSK9 inhibition, lipid metabolism, and cardiovascular disease treatment. Below, we highlight five of these references that reflect comparable therapeutic strategies. These examples offer insight into how similar challenges have been addressed across different approaches to lipid control.
#1. BR112014000392A2
This Brazilian patent, BR112014000392A2, published in 2017, presents therapeutic regimens for lowering LDL-C using PCSK9 antagonist antibodies. The invention emphasizes dosing strategies both alone and in combination with statins. It contributes important clinical insights into antibody-based lipid management, supported by robust pharmacokinetic and pharmacodynamic data.
Below, we have added snapshots from the GPS tool highlighting the relevant snippets from the specification for the similar patents.

What This Patent Introduces To The Landscape
- Targeted PCSK9 antibody therapy: Administration of monoclonal antibodies like L1 L3 to inhibit PCSK9 and enhance LDL clearance.
- Flexible dosing regimens: Initial and maintenance doses ranging from 0.25 mg/kg to over 300 mg, adjusted weekly to monthly.
- Statin co-therapy: Regimens often combine antibody therapy with stable statin doses to amplify LDL-C reduction.
- Broad cardiovascular scope: Addresses familial hypercholesterolemia, atherogenic dyslipidemia, and general cardiovascular disease prevention.
- Clinical validation: Includes detailed pharmacokinetic and pharmacodynamic studies confirming LDL-C reduction in human trials.
How It Connects To EP3536712B1
- Both patents target PCSK9 as a pathway to reduce cardiovascular risk.
- EP3536712B1 focuses on Lp(a) levels, while this reference centers on LDL-C reduction.
- Each patent outlines antibody-driven treatments that offer alternatives to standard statin therapy.
Why This Matters
This reference reinforces the clinical relevance of PCSK9 inhibition as a strategy for lipid modulation. It offers a pharmacologically grounded comparison point for EP3536712B1, especially in terms of antibody specificity, dosing variability, and patient stratification based on statin use.
#2. US2014099312A1
This US application, US2014099312A1, published in 2014, discloses fully human monoclonal antibodies and antigen-binding fragments that specifically bind and neutralize human PCSK9. It offers a detailed framework for therapeutic intervention in hypercholesterolemia using antibody-based inhibition, validated through extensive in vivo and in vitro studies.

What This Patent Introduces To The Landscape
- High-affinity anti-PCSK9 antibodies: Describes fully human monoclonal antibodies targeting specific PCSK9 epitopes, including gain-of-function variants.
- Comprehensive efficacy profile: Demonstrates significant reductions in LDL-C (up to 80%), total cholesterol, and triglycerides with sustained effects in animal and human models.
- Epitope-specific binding: Antibodies bind to propeptide or C-terminal domains, showing selective binding characteristics across species and mutations.
- Receptor blocking mechanism: Antibodies effectively block PCSK9’s interaction with LDL receptors (LDLR), restoring LDL uptake in hepatocyte models.
- Dosing and administration strategies: Offers flexible, sequential dosing regimens (e.g., 75 mg and 150 mg Q2W), adaptable to patient response and statin compatibility.
- Broad therapeutic application: Targets patient populations with familial hypercholesterolemia, statin intolerance, and PCSK9 gain-of-function mutations.
- Statin combination potential: Explores use alongside statins, ezetimibe, niacin, bile resins, and other lipid-lowering agents.
- Preclinical validation: Includes robust animal model testing (mice, hamsters, monkeys) to establish dose-dependence and cross-species lipid regulation.
How It Connects To EP3536712B1
- Both patents use fully human antibodies to inhibit PCSK9 and regulate lipid levels.
- US2014099312A1 focuses primarily on LDL-C reduction, while EP3536712B1 targets Lp(a) in a statin-free context.
- Each invention supports biologic-based cholesterol management with potential for statin-independent or complementary therapy.
Why This Matters
This patent adds depth to the therapeutic narrative of PCSK9 inhibition. It provides molecular precision, diverse epitope targeting, and robust dosing insight that collectively support and contextualize the lipid-lowering strategies proposed in EP3536712B1. The rise of AI-driven drug discovery platforms has accelerated the pace of such innovations.
#3. KR20110056317A
This Korean patent application, KR20110056317A, published in 2011, discloses antagonists of PCSK9, including antibodies, peptides, and aptamers. It demonstrates for the first time that certain PCSK9 antagonists, administered in vivo, effectively lower LDL cholesterol and offer therapeutic potential for cardiovascular and lipid metabolism disorders.
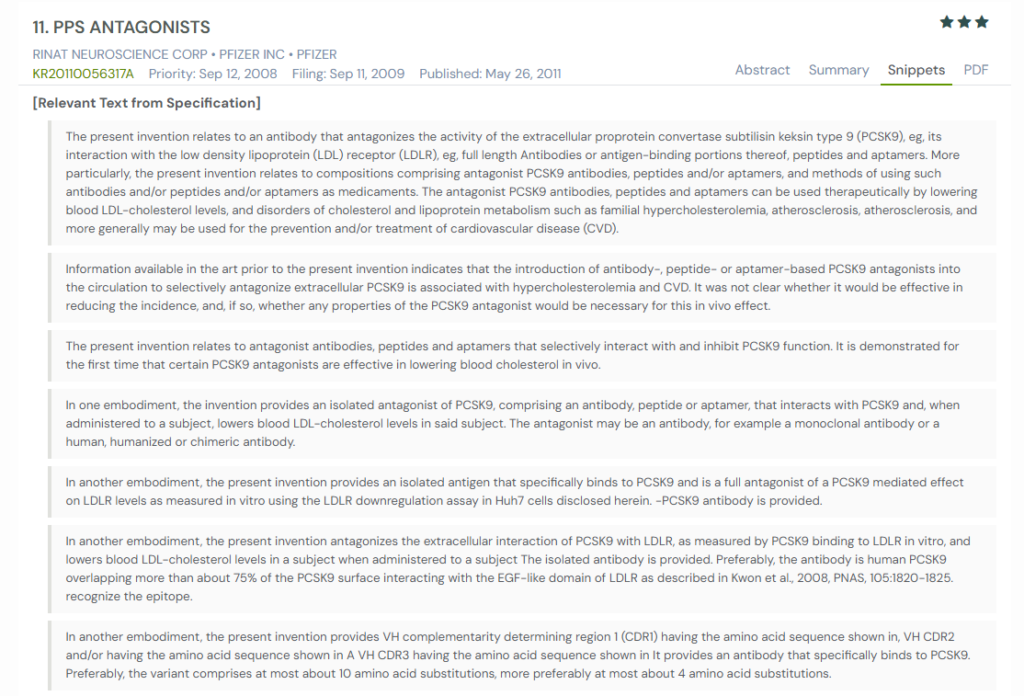
What This Patent Introduces To The Landscape
- Multimodal PCSK9 inhibition: Describes monoclonal antibodies, peptides, and aptamers that block PCSK9 interaction with LDL receptors.
- Cholesterol-lowering efficacy: Demonstrates dose-dependent reductions in LDL-C in mice and cynomolgus monkeys using lead antibodies (e.g., 7D4, 4A5, 6F6).
- Mechanistic specificity: Defines “full antagonist” as agents that block ≥98% of PCSK9 activity in cellular LDLR downregulation assays.
- Epitope-targeted design: Antibodies overlap with regions of PCSK9 that interact with the LDLR EGF-like domain.
- Detailed antibody engineering: Provides sequences for high-affinity CDR variants, engineered through affinity maturation.
- Flexible dosing routes: Covers systemic and localized administration—including intravenous, subcutaneous, and inhalation.
- Broad therapeutic indications: Targets hypercholesterolemia, familial hypercholesterolemia, dyslipidemia, atherosclerosis, and coronary heart disease.
- Combinatorial use: Supports co-administration with other PCSK9 antagonists or lipid-lowering agents.
- In vivo validation: Backed by time- and dose-dependent data showing LDL-C reductions in both preclinical species.
How It Connects To EP3536712B1
- Both patents center on blocking extracellular PCSK9 to improve lipid profiles and reduce cardiovascular risk.
- KR20110056317A supports multiple molecular strategies (antibodies, peptides, aptamers), whereas EP3536712B1 focuses specifically on antibodies.
- Each invention supports statin-independent LDL or Lp(a) modulation through biologic PCSK9 antagonism.
Why This Matters
This reference highlights the diversity of biologic formats used to neutralize PCSK9 and lower cholesterol. It reinforces the therapeutic validity of the pathway targeted by EP3536712B1, while broadening the technological scope through peptide and aptamer-based alternatives. That same thermal-resilience thinking is central to US12245996B2, where formulation pH, chelators, and low-oxygen packaging enable autoclaving and long shelf life for norepinephrine.
#4. KR20100091191A
This Korean patent application, KR20100091191A, published in 2010, presents methods and compositions for treating cholesterol-related disorders using anti-PCSK9 antibodies and antigen-binding fragments. It offers a strategic approach to modulating cholesterol metabolism by directly inhibiting the PCSK9-LDLR interaction. This is a protein-protein interface that has traditionally been viewed as difficult to disrupt with biologics or small molecules.
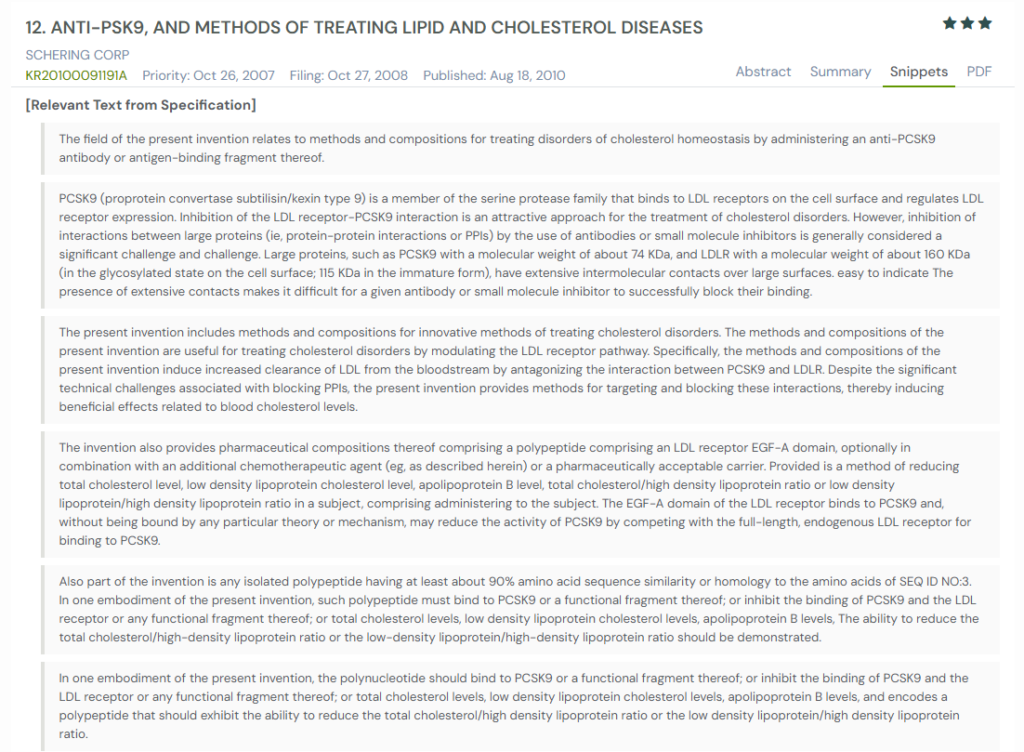
What This Patent Introduces To The Landscape
- Direct PCSK9-LDLR inhibition: Discloses antibodies and EGF-A domain peptides that block the binding interface between PCSK9 and LDL receptors.
- Domain-specific antibodies: Highlights antibodies targeting both the catalytic and C-terminal domains of PCSK9, including epitope-specific binding near amino acids 490–502.
- Use of EGF-A mimetic peptides: Introduces soluble LDLR EGF-A domain mimetics as decoys to competitively inhibit PCSK9.
- Combination therapies: Envisions co-administration with statins, ezetimibe, and cardiovascular drugs for synergistic lipid lowering.
- Cross-species specificity: Describes antibodies that selectively bind to human PCSK9 over mouse or rat homologs.
- In vivo LDL uptake restoration: Demonstrates increased LDL receptor levels and LDL uptake in HepG2 cells and mouse models.
- Potent neutralization profile: Includes IC₅₀ and inhibition rates of multiple lead antibodies (e.g., 1F11/1G11, 75B9, 77D10) with effective cholesterol modulation.
- Multiformat antibodies: Covers monoclonal, polyclonal, camelized, humanized, single-chain, and domain-specific antibody structures.
- Assay-validated results: Supports claims with in vitro receptor-binding assays and in vivo cholesterol modulation experiments in mice.
How It Connects To EP3536712B1
- Both inventions inhibit extracellular PCSK9 to modulate cholesterol and lipoprotein metabolism.
- KR20100091191A expands the inhibition strategy by introducing EGF-A peptide mimics in conjunction with antibodies.
- Each supports the clinical potential of PCSK9 blockade as an independent or adjunct lipid-lowering therapy.
Why This Matters
This reference strengthens the mechanistic foundation for PCSK9 inhibition by combining epitope-level antibody design with receptor mimicry. It serves as a compelling counterpart to EP3536712B1, confirming the viability of disrupting PCSK9-LDLR interactions for broad lipid disorder treatment.
#5. US10023654B2
This U.S. patent, US10023654B2, published in 2018, describes fully human anti-PCSK9 monoclonal antibodies and their use in treating cholesterol-related disorders. It introduces potent neutralizing antibodies with high specificity and durability in reducing LDL cholesterol and total cholesterol levels across multiple species and model systems.
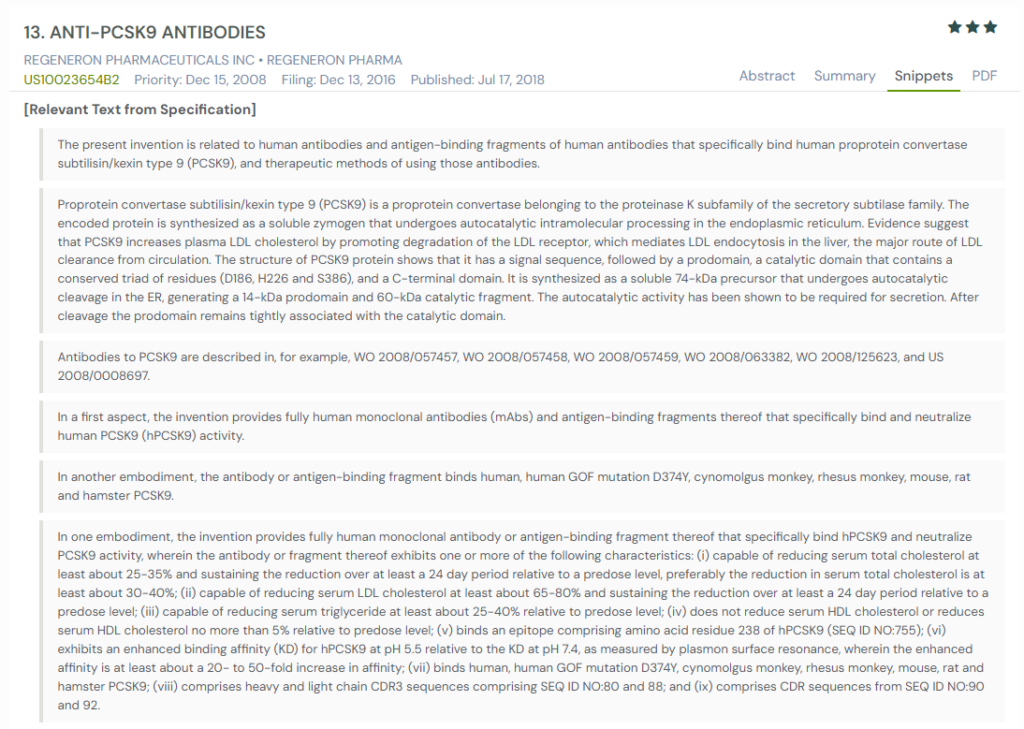
What This Patent Introduces To The Landscape
- Fully human monoclonal antibodies: Describes mAbs and fragments that specifically bind and neutralize human PCSK9 activity.
- Cross-species targeting and selectivity: Includes antibodies with binding profiles across human, monkey, and select animal PCSK9 variants.
- Robust LDL-C and total cholesterol reduction: Reports LDL-C suppression of up to 80%, sustained for over 90 days depending on dose and antibody.
- Epitope-specific binding: Targets distinct regions within the PCSK9 catalytic and prodomain regions.
- Acidic pH binding enhancement: Some antibodies exhibit increased affinity at acidic pH, favoring endosomal efficacy.
- ELISA and surface plasmon validation: Includes extensive binding assays and IC50 data for LDLR interaction blocking.
- Pharmacokinetic profiling: Reports long-acting serum stability and therapeutic efficacy in PCSK9-humanized mouse and hamster models.
- Combination therapy compatibility: Designed for co-administration with statins, niacin, and other lipid-lowering drugs.
- Multispecific constructs: Covers formats including monospecific, bispecific, and engineered multispecific antibodies.
How It Connects To EP3536712B1
- Both patents focus on using antibodies to inhibit PCSK9, thereby reducing LDL-C and improving lipid profiles.
- US10023654B2 includes more structural antibody insights, while EP3536712B1 centers on therapeutic application.
- Each validates PCSK9 as a high-impact target for cardiovascular and cholesterol-related treatment strategies.
Why This Matters
This work reinforces the clinical viability of anti-PCSK9 monoclonal antibodies as a leading approach in lipid-lowering therapies. With detailed antibody engineering and pharmacodynamic validation, US10023654B2 complements EP3536712B1 by anchoring the therapeutic framework in robust biochemical data.
How To Find Related Patents Using Global Patent Search
Understanding the broader patent landscape is crucial when working on lipid metabolism, cholesterol regulation, or monoclonal antibody therapies. The Global Patent Search tool simplifies this task by surfacing inventions that explore similar biological targets, such as PCSK9, and their therapeutic applications.
1. Enter the patent number into GPS: Start by inputting a patent number like EP3536712B1 into the GPS tool. The platform converts it into a smart search that you can narrow with terms like “anti-PCSK9”, “lipid metabolism”, or “LDL receptor inhibition”.
2. Explore conceptual snippets: Rather than line-by-line claim comparisons, GPS now delivers curated text snippets. These showcase how other inventions approach PCSK9 inhibition, antibody design, or cholesterol clearance mechanisms.
3. Identify related inventions: The tool uncovers patents that aim to lower LDL-C levels, block PCSK9 activity, or manage hypercholesterolemia, helping users track innovations across therapeutic classes.
4. Compare biological mechanisms, not just claims: GPS focuses on functional overlaps rather than legal phrasing. This highlights how different systems achieve similar lipid-lowering outcomes or antibody responses.
5. Accelerate cross-domain insights: Whether you’re researching cardiovascular therapies, monoclonal antibody engineering, or cholesterol metabolism, GPS helps uncover parallel ideas that might otherwise be overlooked.
By revealing how similar therapeutic ideas have been patented, Global Patent Search helps both legal and scientific teams make informed, strategic decisions.
Disclaimer: The information provided in this article is for informational purposes only and should not be considered legal advice. The related patent references mentioned are preliminary results from the Global Patent Search tool and do not guarantee legal significance. For a comprehensive related patent analysis, we recommend conducting a detailed search using GPS or consulting a patent attorney.




