Some patents unlock niche advances. Others, like European patent EP2590626B1, drive global shifts in vaccine delivery and RNA-based immunization.
Assigned to GlaxoSmithKline Biologicals SA, this patent outlines a liposomal, non-viral system for delivering RNA encoding immunogens. Its lipid bilayer design protects RNA and enhances cellular translation, a key feature in next-generation vaccine platforms.
The patent has drawn international attention, with ongoing disputes involving Moderna in multiple jurisdictions. However, this article focuses on the science, rather than the courtroom.
Using the Global Patent Search (GPS) tool, we explore patents with overlapping design elements, like lipid composition, RNA encapsulation, and self-replicating mechanisms.
Whether you’re building an IP strategy or tracking innovation in immunology, this GPS-driven review offers critical insight into the landscape surrounding EP2590626B1.
Understanding Patent EP2590626B1
European patent EP2590626B1, assigned to GlaxoSmithKline Biologicals SA, discloses a non-viral delivery system for RNA-based immunization. The invention uses liposomes containing lipids with a pKa of 5.0 to 6.8 to encapsulate RNA that encodes immunogens.
These specially engineered liposomes protect the RNA from degradation and enable efficient delivery to vertebrate cells. Once inside, the RNA is translated to produce immunogenic proteins, triggering an immune response.

Source: Google Patents
Its Four Key Features Are
#1. pKa-optimized lipid composition: The lipid bilayer of the liposome includes at least one lipid with a pKa between 5.0 and 6.8, often featuring a tertiary amine.
#2. Encapsulation of immunogen-encoding RNA: The aqueous core of the liposome contains single-stranded RNA that encodes a desired immunogen, such as RSV F protein or others.
#3. Self-replicating RNA capability: The RNA may be a replicon, encoding its own RNA polymerase, enabling in situ amplification and protein expression.
#4. Efficient immune activation: The liposomes promote cellular uptake and immune stimulation, resulting in a strong antibody and T-cell response.
This technology supports high-efficiency RNA delivery without the use of viral vectors. It forms a foundation for next-generation RNA vaccine platforms.
Note: Durability is vital for vaccines and delivery. EP2279254B1 Patent Audit echoes how RNA vaccine platforms rely on stability to survive circulation and enhance immune response.
Similar Patents As EP2590626B1
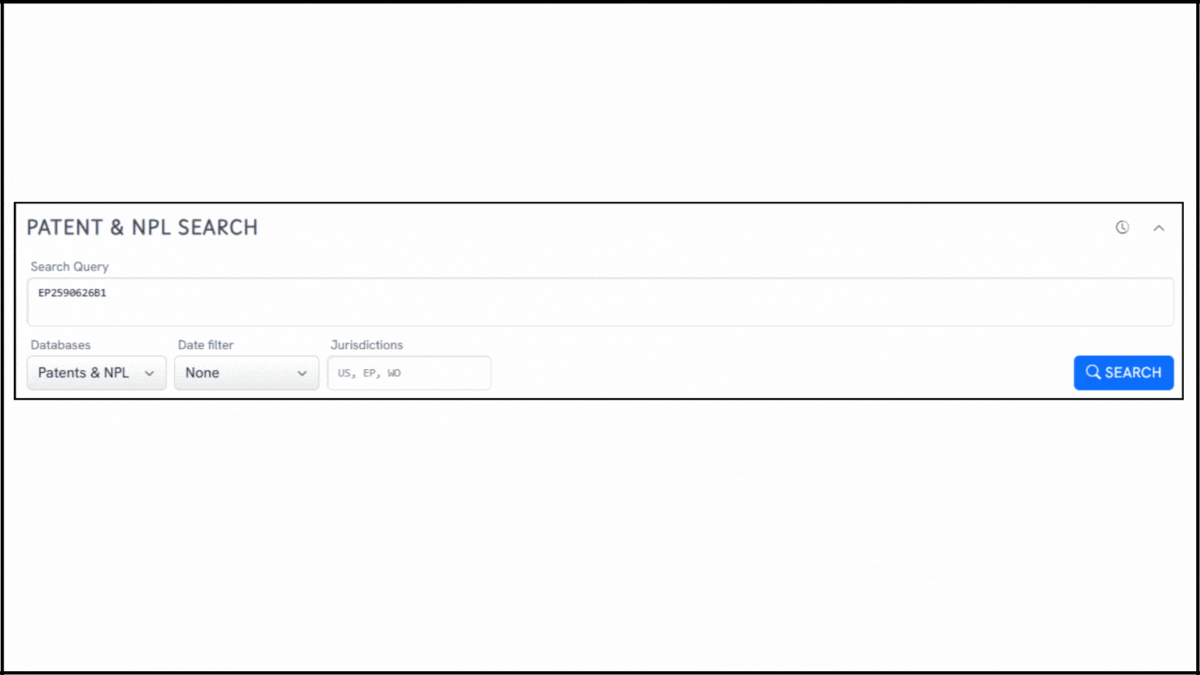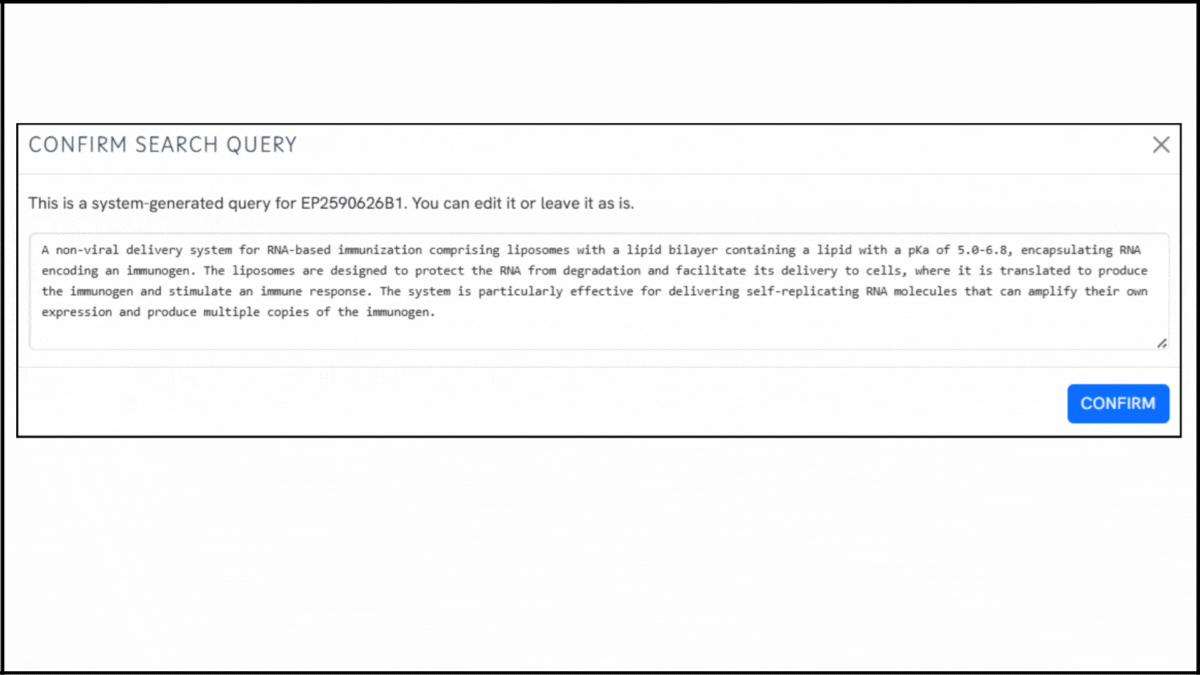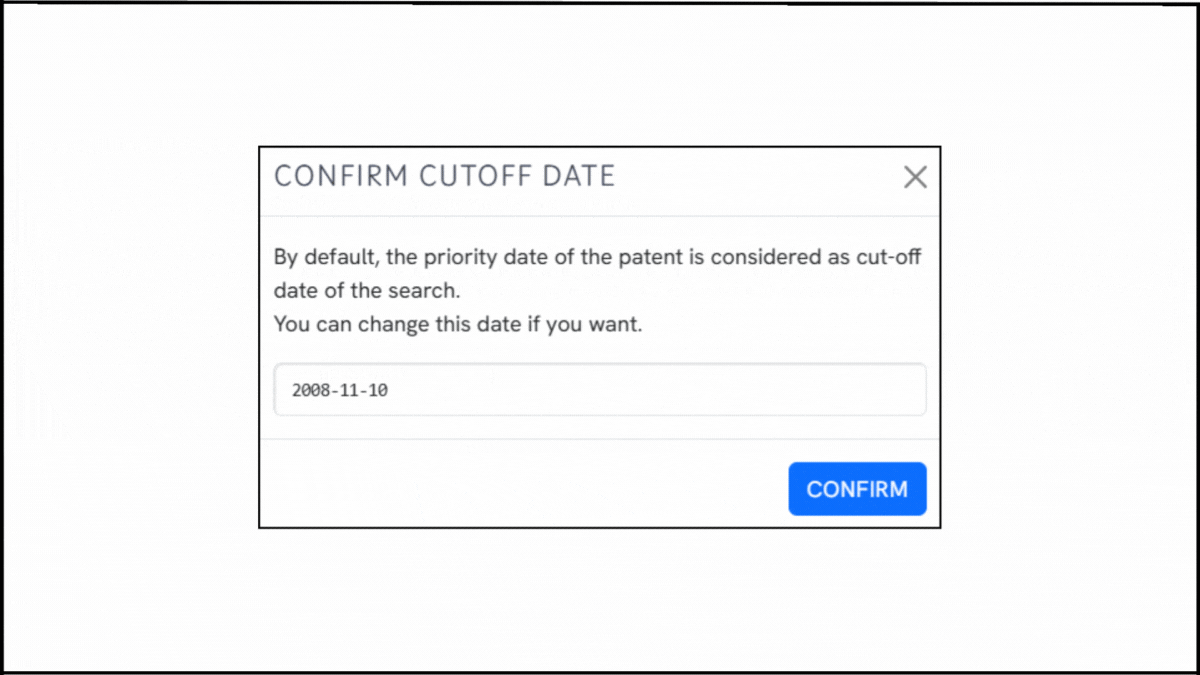
To explore the innovation landscape surrounding EP2590626B1, we ran the patent through the Global Patent Search tool. Below is a quick glimpse of the GPS tool in action:
Source: Global Patent Search
This analysis surfaced a set of patents that reflect similar concepts in liposomal RNA delivery, immunogen encoding, and immune response triggering. These are core themes in today’s AI-driven healthcare innovations. Below, we highlight five key references that contribute to this evolving field of RNA-based vaccine platforms.
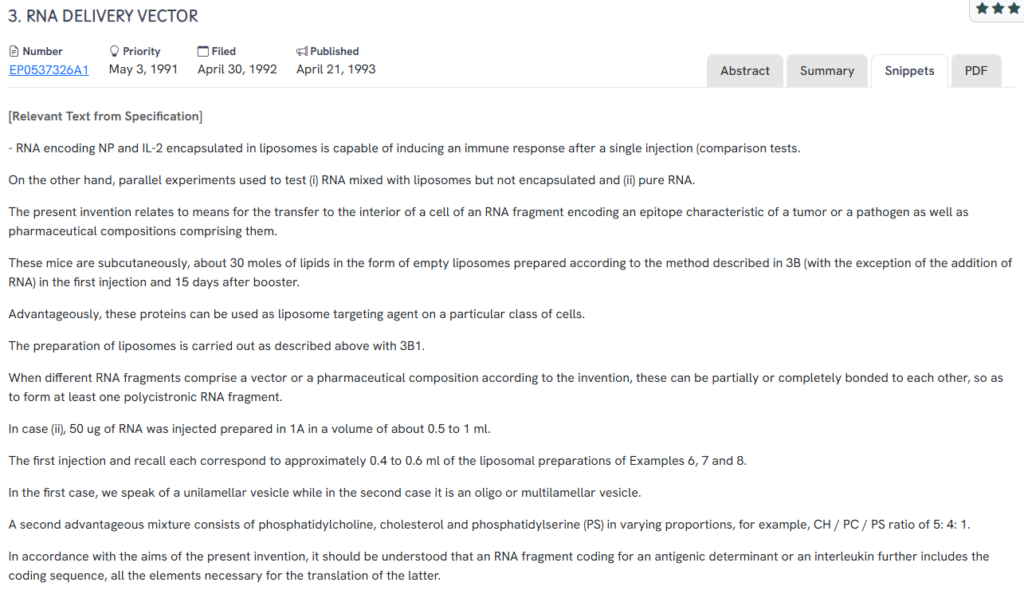
#1. EP0537326A1
This European patent, EP0537326A1, published in 1993, presents liposome-encapsulated RNA compositions for vaccine delivery targeting tumor or pathogen-related epitopes. It focuses on subcutaneous or intramuscular administration to induce strong immune responses using RNA-lipid formulations.
Below, we have added snapshots from the GPS tool highlighting the relevant snippets from the specification for the similar patents.
What This Patent Introduces To The Landscape
- Encapsulation of RNA in liposomes: Demonstrates immunogenic response after single-dose administration using RNA encoding NP and IL-2.
- Unilamellar and multilamellar liposomes: Explores different vesicle structures for delivering RNA directly into host cells.
- Customizable lipid compositions: Uses phosphatidylcholine, cholesterol, and phosphatidylserine in varying ratios for targeted delivery.
How It Connects To EP2590626B1
- Both encapsulate immunogen-encoding RNA to trigger immune responses.
- Each relies on liposomal vesicles for RNA stability and cellular delivery.
- Both aim to improve antigen expression without viral vectors.
Why This Matters
This early disclosure shows foundational work in non-viral RNA delivery using liposomes. It supports EP2590626B1’s approach to immune system activation via RNA-lipid formulations.
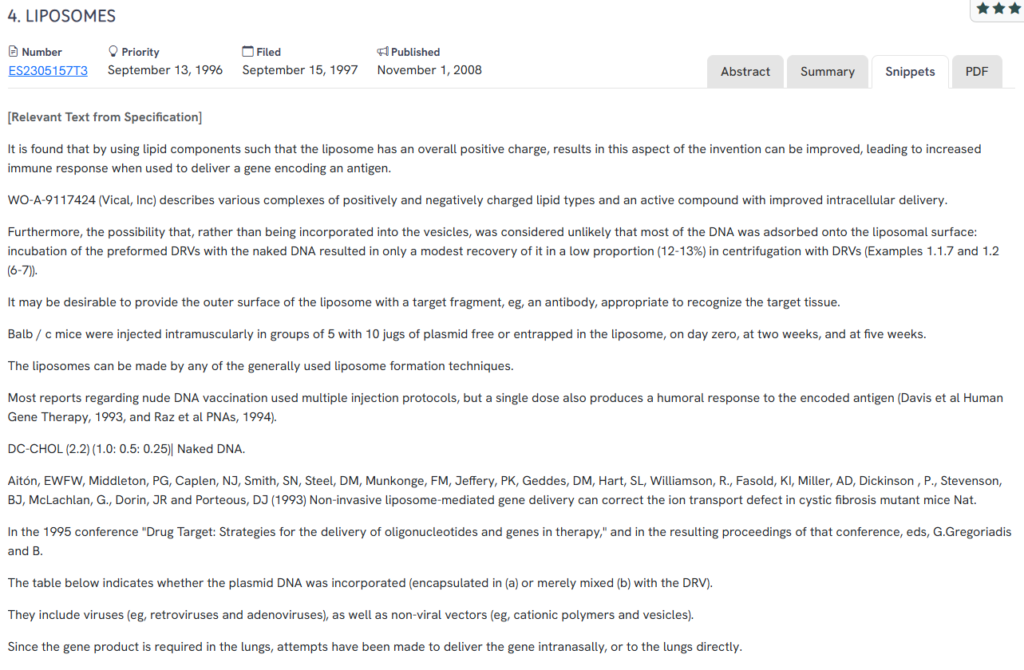
#2. ES2305157T3
This Spanish patent, ES2305157T3, published in 2008, explores positively charged liposomes as vehicles for delivering genes encoding antigens. It emphasizes that surface charge plays a key role in enhancing immunogenicity during gene delivery.
What This Patent Introduces To The Landscape
- Cationic lipid composition: Shows that positively charged liposomes enhance immune response when delivering genetic material.
- Surface-targeting capability: Suggests attaching antibodies to the liposome exterior to target specific tissues.
- DNA encapsulation efficacy: Differentiates between entrapped DNA and DNA merely adsorbed to the vesicle surface.
How It Connects To EP2590626B1
- Both focus on non-viral delivery systems using lipid-based vesicles.
- Each emphasizes the role of lipid chemistry in delivery and immune activation.
- Both discuss the encapsulation of nucleic acids for enhanced expression and stability.
Why This Matters
This patent highlights how the liposomal surface charge and structural formulation influence the immune response, core principles also evident in EP2590626B1’s RNA vaccine strategy.
Did you know: Durability is as critical for vaccines as it is for peptide drugs. EP3175863B1 and Similar Patents on Extended-Action Peptide Drugs highlight PEGylation strategies that mirror stability improvements seen in RNA-based therapies.
#3. CA2700808A1
This Canadian patent, CA2700808A1, published in 2009, presents a delivery system using liposomes suspended in a continuous hydrophobic phase for in vivo polynucleotide delivery. It highlights siRNA and DNA vaccine combinations to boost immune response and therapeutic impact.

What This Patent Introduces To The Landscape
- Hydrophobic carrier enhancement: Demonstrates improved in vivo delivery of siRNA using a liposome-in-oil carrier system.
- Immune activation via RNA-based adjuvants: Describes how internalized polynucleotides interact with intracellular receptors to activate immune pathways.
- Multilamellar liposome formulation: Utilizes phospholipid and cholesterol blends to encapsulate DNA and siRNA effectively.
How It Connects To EP2590626B1
- Both use liposomal formulations to protect and deliver RNA or DNA-based immunogenic material.
- Each system improves antigen-specific immune response through intracellular delivery.
- Both consider structural lipid design to be crucial for delivery efficacy.
Why This Matters
This patent showcases an innovative lipid-based RNA delivery method that aligns with EP2590626B1’s goal of enhancing immune stimulation using stabilized, non-viral vectors.
#4. ES2646669T3
This Spanish patent, ES2646669T3, published in 2017, details the use of liposome-encapsulated, self-replicating RNA to enhance immune responses. It compares favorably to virion-based delivery methods, emphasizing efficient intracellular translation and antigen expression.

What This Patent Introduces To The Landscape
- Self-replicating RNA delivery: Uses replicon RNA that encodes its own polymerase for amplified antigen production after delivery.
- Liposome encapsulation for expression: Confirms encapsulation is critical for effective RNA translation and strong immune activation.
- Targeted immune modulation: Combines RNA with TLR agonists to stimulate dendritic cells and enhance cytokine secretion.
How It Connects To EP2590626B1
- Both deliver RNA via liposomes to promote robust expression of encoded immunogens.
- Each emphasizes the use of replicon-based RNA for self-amplifying antigen output.
- Both demonstrate strong, dose-efficient immune responses through lipid-formulated RNA delivery.
Why This Matters
This patent confirms that liposomal encapsulation of replicating RNA is essential for effective immune engagement, a core principle also advancing in synthetic biology innovation, and directly supporting EP2590626B1’s RNA delivery strategy.
#5. CA3169291A1
This Canadian patent, CA3169291A1, published in 2012, presents a liposomal RNA delivery method optimized for immunizing large mammals. It emphasizes low-dose delivery of self-replicating RNA to trigger strong immune responses through in situ antigen expression.

What This Patent Introduces To The Landscape
- Encapsulation of replicon RNA: Confirms liposome-encapsulated RNA induces immune responses comparable to virion-based delivery systems.
- Lipid formulation for large animals: Uses a precise blend of cationic, zwitterionic, and PEGylated lipids for effective delivery.
- Low-dose immunization: Demonstrates robust immunity in large mammals with RNA doses as low as 2 μg per administration.
How It Connects To EP2590626B1
- Both rely on self-replicating RNA encapsulated in liposomes to boost immune responses.
- Each highlights the necessity of encapsulation for effective translation and antigen presentation.
- Both target systemic and mucosal immunity using non-viral delivery strategies.
Why This Matters
This patent highlights how lipid-encapsulated replicon RNA can induce immunity at low doses, aligning with the goals of EP2590626B1 in developing efficient and scalable RNA vaccine designs.
How To Find Related Patents Using Global Patent Search
Understanding the broader patent landscape is essential when working with RNA-based immunization, lipid delivery systems, or vaccine formulation. The Global Patent Search tool streamlines this task, enabling users to uncover technologies that align with or parallel RNA-liposome delivery strategies.
1. Enter the patent number into GPS: Begin by entering EP2590626B1 into the GPS tool. It automatically generates a smart query, which you can refine using terms like lipid encapsulation, replicon RNA, or immunogen delivery.
2. Explore conceptual snippets: Rather than mapping features line-by-line, GPS now delivers curated snippets. These reveal how other inventions approach RNA stability, immune activation, or intracellular delivery.
3. Identify related inventions: The tool surfaces patents that use cationic lipids, RNA replicons, or nanoparticle systems, offering insight into how similar biological challenges have been addressed.
4. Compare systems, not legal claims: Instead of parsing claim language, GPS compares functional intent: how RNA is protected, translated, or targeted, enabling quicker understanding of shared mechanisms.
5. Accelerate cross-domain insights: Whether you’re focused on vaccine engineering, delivery vectors, or immune system design, GPS helps reveal connections across biopharma and genetic medicine domains. Biomedical innovation stretches from RNA-based therapies to structural biomaterials like the variable-denier sutures in EP3835470B1.
With this concept-first approach, Global Patent Search empowers researchers, IP teams, and developers to track innovation in immunology or AI-driven drug discovery with speed, clarity, and technical precision.
Disclaimer: The information provided in this article is for informational purposes only and should not be considered legal advice. The related patent references mentioned are preliminary results from the Global Patent Search tool and do not guarantee legal significance. For a comprehensive related patent analysis, we recommend conducting a detailed search using GPS or consulting a patent attorney.




