When medicines are designed to last longer in the body, even small changes can make a big difference. EP3175863B1 is now part of a revocation proceeding involving Ascendis Pharma and BioMarin Pharmaceutical Inc.
This European patent focuses on new versions of a natural peptide called C-type natriuretic peptide (CNP). These versions are made to stay active in the bloodstream for longer, making treatments more effective with fewer doses. Such advances could help patients with conditions like achondroplasia by helping the therapy work better for a longer time.
To explore related inventions, we use the Global Patent Search (GPS) platform. GPS helps uncover similar patents and track new developments across medical research.
Whether you are a student, researcher, or simply curious about medical innovation, this GPS-driven review highlights the ideas behind EP3175863B1.
Understanding Patent EP3175863B1
EP3175863B1 covers variants of C-type natriuretic peptide (CNP) that are designed to last longer in the bloodstream. The goal is to slow down how quickly the body breaks them down, while preserving their natural therapeutic function. These variants are made through modifications such as PEGylation, amino acid substitutions, and N-terminal extensions, allowing the therapy to remain effective with lower and less frequent doses.

Source: Google Patents
Its Four Key Features Are
#1. Increased serum half-life: The CNP variants are engineered to remain active in the bloodstream for longer compared to natural CNP.
#2. PEGylation for protection: PEG chains are attached to the peptide to prevent degradation by enzymes such as neutral endopeptidase (NEP).
#3. Structural modifications: Amino acid substitutions and N-terminal extensions are introduced to further improve stability and maintain biological activity.
#4. Optimized molecular size: The PEG-CNP variants have masses ranging from 2.6 to 7 kDa, which increases resistance to enzymatic cleavage and clearance.
This patent is significant because it focuses on enhancing CNP-based therapies for bone disorders, such as achondroplasia, and vascular smooth muscle disorders. By extending the half-life of CNP, the therapy can be given at lower doses with longer-lasting effects.
Fun fact: Drug-delivery patents echo similar goals across industries. US10874703B2 and similar patents refine botanical infusion by using heat and pressure to ensure even compound distribution, comparable to controlled-release strategies in pharmaceuticals.
Similar Patents As EP3175863B1
To explore the innovation landscape surrounding EP3175863B1, we ran the patent through the Global Patent Search tool. This surfaced related patents that share technical similarities in peptide modification, PEGylation strategies, and durability enhancement.
Below, we highlight five of these references that reflect comparable ideas in extending peptide stability and therapeutic performance.
#1. JPH03148298A
This Japanese patent, JPH03148298A, published in 1991, describes a method for attaching polyethylene glycol (PEG) derivatives to peptides. The modification improves their durability in the bloodstream, reduces clearance, and preserves activity. It also outlines manufacturing techniques that allow control over the degree of PEGylation by adjusting reaction conditions.
Below, we have added snapshots from the GPS tool highlighting the relevant snippets from the specification for the similar patents.
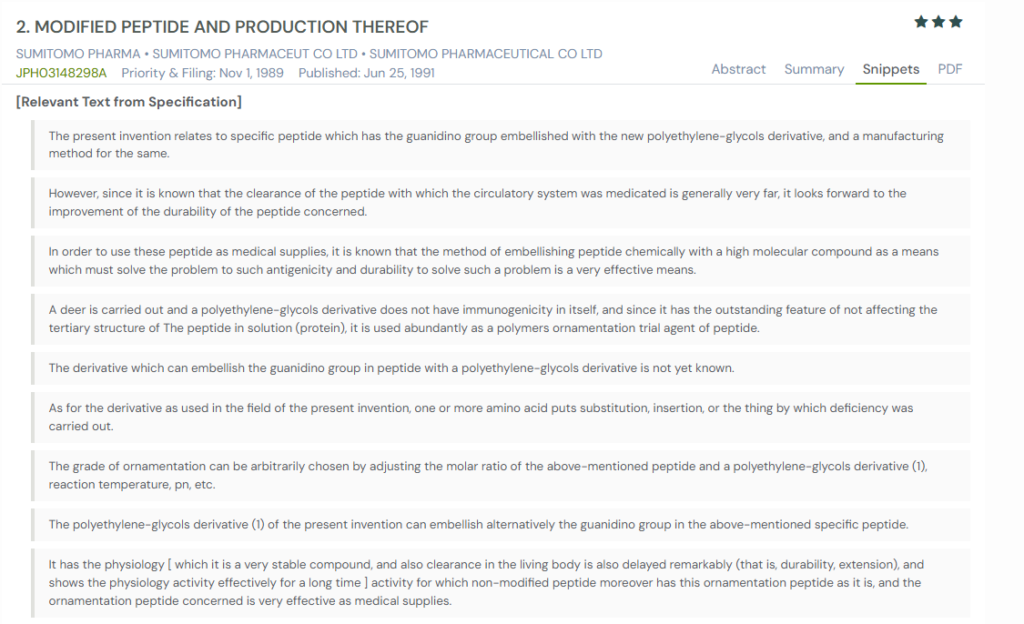
What This Patent Introduces To The Landscape
- PEGylation of guanidino groups – Enhances peptide stability and delays clearance from the body.
- Controlled modification process – Reaction parameters can be adjusted to fine-tune the level of PEG attachment.
- Non-immunogenic PEG derivatives – PEG chains used do not trigger immune reactions and maintain peptide structure in solution.
- Extended biological activity – Modified peptides show longer-lasting therapeutic effects compared to unmodified forms.
How It Connects To EP3175863B1
- Both patents focus on PEGylation as a means to extend peptide half-life.
- Each aims to overcome rapid clearance and degradation in the body.
- Both emphasize maintaining biological activity while improving therapeutic durability.
Why This Matters
This reference shows how PEGylation was already being applied to improve peptide medicines decades earlier. It illustrates the foundation of durability-focused drug design, which EP3175863B1 builds upon with more advanced peptide engineering. Together, they highlight a clear trajectory in therapeutic peptide development.
You may also want to explore how formulation claims are defended in litigation, such as the recent case involving Allergan’s US12097187B2.
#2. US2007184015A1
This US patent application, US2007184015A1, published in 2007, introduces a novel branched PEG design for extending the half-life of therapeutic proteins and peptides. This invention discloses a three-arm PEG molecule with a single attachment point, designed to provide a stronger protective shield against enzymatic degradation. It also emphasizes that the PEG constructs are biodegradable, ensuring that the therapeutic agent gradually breaks down in the body after exerting its effect.
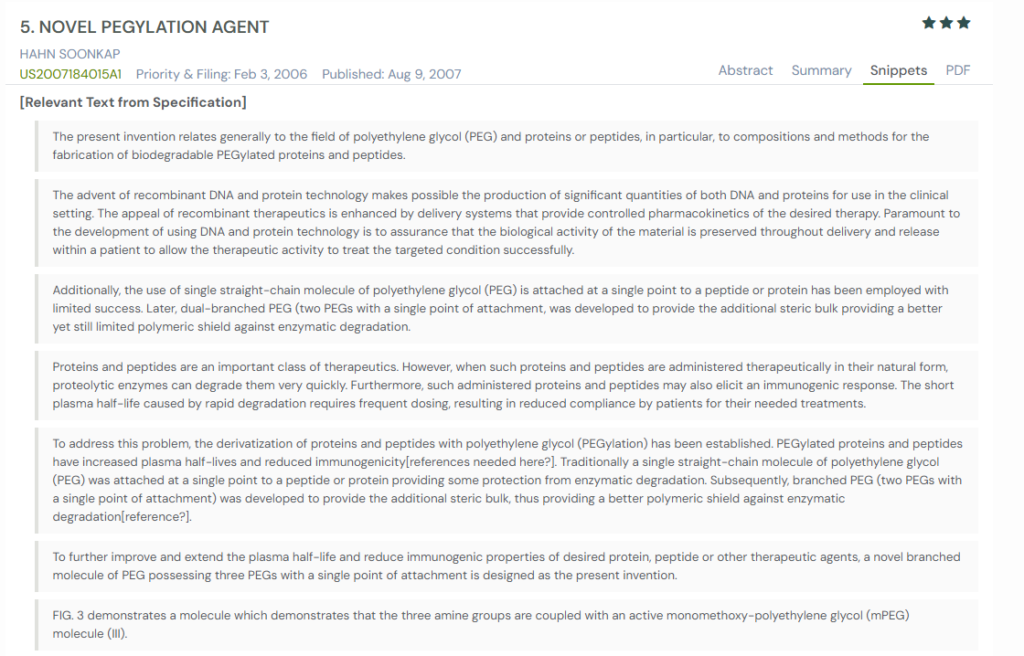
What This Patent Introduces To The Landscape
- Tri-branched PEG molecule – Three PEG arms attached at a single point provide greater steric protection than straight or dual-branched PEG designs.
- Enhanced plasma half-life – Modified proteins and peptides resist enzymatic degradation, significantly extending circulation time in the body.
- Reduced immunogenicity – PEGylation decreases the likelihood of immune reactions against the therapeutic molecule.
- Biodegradable PEG composites – Designed to degrade gradually by enzymatic or hydrolytic processes for safe clearance.
How It Connects To EP3175863B1
- Both patents aim to prolong peptide half-life through PEGylation strategies.
- Each addresses the problem of rapid enzymatic degradation and short plasma persistence.
- Both emphasize maintaining therapeutic activity while reducing immune response.
Why This Matters
This reference highlights an important step forward in PEGylation technology, moving from single and dual branches to a tri-branched system. It reflects the continuous refinement of PEG-based drug delivery, showing how structural variations can improve durability and patient compliance. Together with EP3175863B1, it demonstrates the evolution of peptide PEGylation toward more effective and safer therapies.
This emphasis on long-term formulation reliability is also central to US10869845B1, which demonstrates how injectable ephedrine can remain stable without repeated preparation or dilution.
#3. CA2017541A1
This Canadian patent application, CA2017541A1, published in 1990, discloses novel polyethylene glycol (PEG) derivatives designed to modify peptides at guanidino groups. The invention emphasizes the selective modification of peptides to improve durability and reduce immunogenicity without disrupting their function. It further outlines manufacturing methods and pharmaceutical compositions, highlighting applications in therapeutic peptides such as GM-CSF, erythropoietin, and atrial natriuretic peptide.
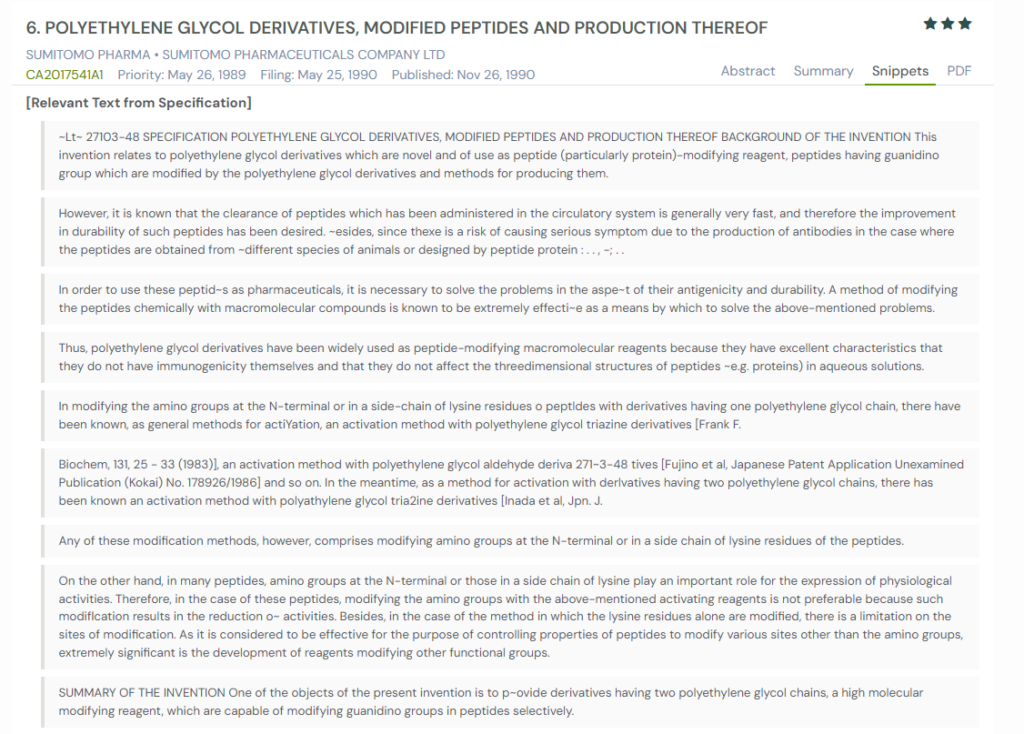
What This Patent Introduces To The Landscape
- Selective guanidino modification – PEG derivatives selectively attach to guanidino groups in peptides to improve stability.
- Improved durability – Modified peptides show delayed clearance, allowing longer-lasting therapeutic activity.
- Reduced antigenicity – PEGylation reduces immune responses compared to unmodified or animal-derived peptides.
- Pharmaceutical applications – Applicable to a wide range of therapeutic peptides, including growth factors and natriuretic peptides.
How It Connects To EP3175863B1
- Both focus on PEGylation to extend peptide half-life in circulation.
- Each emphasizes retaining biological activity while reducing degradation and clearance.
- Both highlight the role of PEG derivatives in creating more durable peptide-based therapies.
Why This Matters
This reference demonstrates early development of PEG derivatives tailored for selective modification of peptides. By focusing on guanidino groups, it expands the modification toolkit beyond N-terminal and lysine residues. Together with EP3175863B1, it illustrates how peptide engineering evolved to improve pharmacological performance, paving the way for longer-lasting peptide medicines.
#4. US5214131A
This US patent, US5214131A, published in 1993, discloses polyethylene glycol (PEG) derivatives designed to modify guanidino groups in peptides selectively. The invention aims to address the challenges of rapid peptide clearance and immunogenicity by introducing stable PEG conjugates that extend durability without compromising biological activity. It also covers methods of production, purification techniques, and applications in a wide range of therapeutic peptides, including hormones, enzymes, and growth factors.

What This Patent Introduces To The Landscape
- Selective guanidino PEGylation – PEG derivatives attach specifically to guanidino groups, expanding modification beyond N-terminal and lysine residues.
- Extended peptide stability – Modified peptides show significant delays in clearance while retaining their physiological activity.
- Broad therapeutic applicability – Targets hormones, enzymes, and growth factors, including insulin, atrial natriuretic peptide, and erythropoietin.
- Optimized production methods – Details variable reaction conditions for tailoring modification levels and purification approaches.
How It Connects To EP3175863B1
- Both patents use PEGylation to enhance peptide half-life and therapeutic durability.
- Each emphasizes retaining biological activity while reducing degradation and clearance.
- Both reflect PEG’s role in enabling safer, longer-lasting peptide-based medicines.
Why This Matters
This reference underscores the importance of selective PEGylation as a strategy to overcome rapid clearance and immune concerns in peptide drugs. By broadening the range of therapeutic peptides that can be effectively modified it highlights a step toward versatile, long-lasting therapies. Together with EP3175863B1, it demonstrates how refinement in PEGylation approaches continues to shape modern biopharmaceutical development.
Whether you’re analyzing long‑acting peptide therapies or opioid formulations for gut disorders, GPS offers structured comparison, for instance, in our detailed review of US12097187B2 and its safety-driven patent family.
Note: Oncology patents like US12178816B2, which use solid dispersion to boost solubility, mirror how peptide drug patents extend action through formulation engineering.
#5. JPS61178926A
This Japanese patent, JPS61178926A, published in 1986, focuses on chemically modifying peptide hormones to delay clearance and reduce antigenicity while preserving biological activity. The invention employs polyethylene glycol derivatives, particularly monomethyl ether PEG, to improve the pharmacological profile of peptide hormones with molecular weights typically between 850 and 8,000. It demonstrates how controlled modification can create safer, longer-lasting peptide therapeutics suitable for diverse physiological functions.
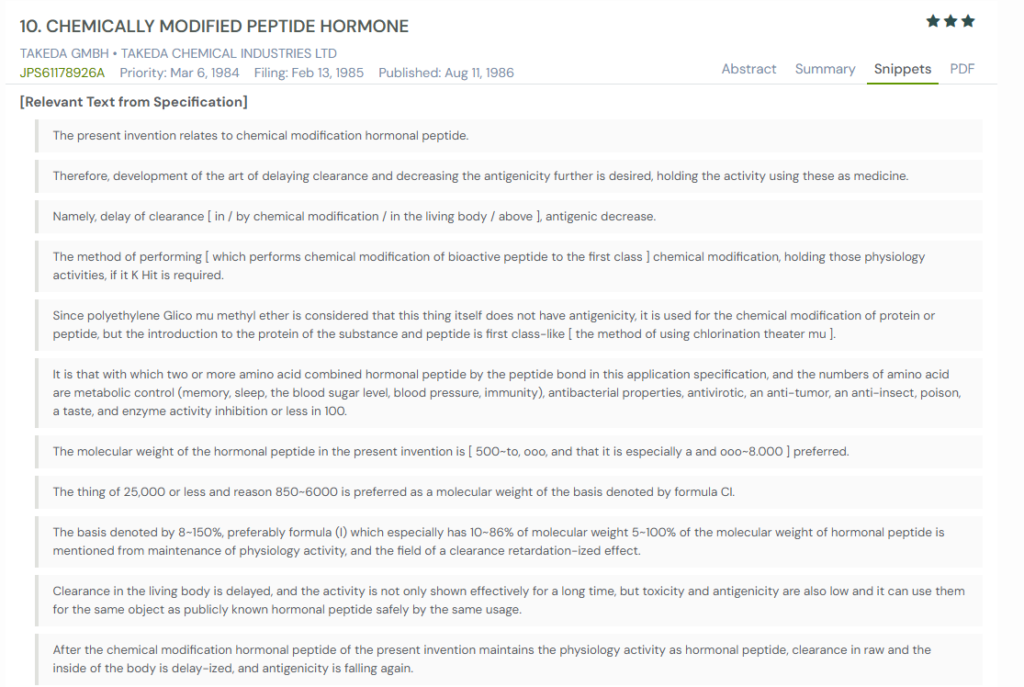
What This Patent Introduces To The Landscape
- PEGylation of peptide hormones – Uses PEG derivatives to extend circulation time while maintaining activity.
- Reduced antigenicity – Modified peptides trigger fewer immune responses compared to unmodified versions.
- Retention of activity – Biological functions such as metabolic control and immunity remain intact after modification.
- Broader safety profile – Modified peptides show reduced toxicity, allowing safer therapeutic applications.
How It Connects To EP3175863B1
- Both patents address the challenge of rapid clearance and immune response in therapeutic peptides.
- Each relies on PEG modification strategies to extend activity without compromising function.
- Both highlight durability as a cornerstone of effective peptide-based drug design.
Why This Matters
This reference illustrates one of the early attempts to optimize peptide hormones through PEGylation for safer, longer therapeutic use. It highlights the importance of durability, reduced antigenicity, and preserved activity as essential goals in peptide drug development. When viewed alongside EP3175863B1, it reveals a clear lineage of innovation, progressing from foundational modifications to modern, highly optimized therapeutic peptides.
Recommended Read: US12144810B1, with its focus on PARP inhibitor solid dispersions, aligns with how extended-action peptide drug patents are pushing drug delivery toward more stable, bioavailable therapies.
How to Find Related Patents Using Global Patent Search
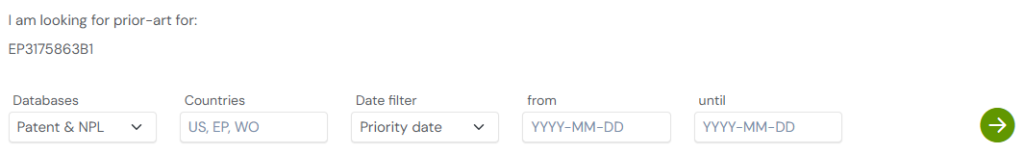
Understanding the broader patent landscape is essential when exploring peptide drugs, PEGylation strategies, or AI-driven healthcare innovations. The Global Patent Search tool makes this easier by surfacing inventions that tackle similar challenges in therapeutic durability and peptide modification.
1. Enter the patent number into GPS: Start by entering a patent number like EP3175863B1 into the GPS tool. The platform transforms it into a targeted query, which can be refined with terms like “PEGylation,” “half-life extension,” or “modified peptides.”
2. Explore conceptual snippets: Instead of comparing features claim-by-claim, GPS now presents curated text snippets. These highlight how other inventions modify peptides, delay clearance, or improve therapeutic activity.
3. Identify related inventions: The tool reveals patents addressing durability, stability, and reduced antigenicity in peptides, offering insight into how similar problems have been solved.
4. Compare systems, not legal claims: Rather than focusing strictly on legal claim language, GPS emphasizes conceptual approaches. This helps users recognize overlaps in strategies for modifying therapeutic peptides.
5. Accelerate cross-domain insights: Whether working in drug delivery, protein engineering, or pharmaceutical innovation, GPS lets researchers uncover related approaches across domains that might otherwise remain disconnected, including the fast-growing field of AI-driven drug discovery.
With Global Patent Search, users can move beyond scattered documents and gain a clear, connected view of how biopharmaceutical innovations build on one another. This clarity supports smarter research decisions, guiding both scientific development and strategic IP planning.
Disclaimer: The information provided in this article is for informational purposes only and should not be considered legal advice. The related patent references mentioned are preliminary results from the Global Patent Search tool and do not guarantee legal significance. For a comprehensive related patent analysis, we recommend conducting a detailed search using GPS or consulting a patent attorney.




