Lipid nanoparticles (LNPs) have become essential to gene therapy and RNA-based treatments. EP2279254B1 plays a central role in this technology shift.
Filed by Arbutus Biopharma, the patent discloses a stable formulation of cationic, non-cationic, and conjugated lipids. It addresses key delivery issues, including toxicity, poor serum stability, and low gene expression.
Now under revocation proceedings involving Moderna and its affiliates across multiple European jurisdictions, the patent has drawn renewed attention within the IP and biotech communities.
But legal questions aside, how unique is this formulation? How many other patents disclose similar delivery structures?
Using the Global Patent Search (GPS) tool, we explore patents that align with EP2279254B1 at the feature level. GPS reveals technical parallels, not just keyword matches, helping professionals find relevant, similar patents faster and with greater precision.
This article explores related innovations that are shaping the future of nucleic acid delivery.
Understanding Patent EP2279254B1
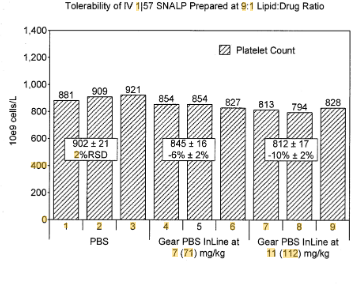
EP2279254B1 describes a stable lipid nanoparticle formulation suitable for the in vivo delivery of nucleic acids, including small interfering RNA (siRNA). The formulation addresses key problems seen in earlier lipid-based delivery systems, particularly toxicity, poor stability in serum, and reduced gene expression. These lipid particles, often referred to as SNALPs (stable nucleic acid-lipid particles), enhance therapeutic outcomes by improving stability, encapsulation, and tolerability.
Source: Google Patents
Its Four Key Features Are
#1. Cationic Lipid Ratio: The particle contains 50–85 mol% % cationic lipid to aid in nucleic acid complexation and delivery.
#2. Non-Cationic Lipid Ratio: Includes 13–49.5 mol% % non-cationic lipids, typically a mix of phospholipids and cholesterol.
#3. Conjugated Lipid Component: PEG-lipid conjugates make up 0.5–2 mol% % to inhibit particle aggregation and enhance circulation time.
#4. Encapsulated Nucleic Acid: siRNA or other nucleic acids are fully enclosed, ensuring protection from enzymatic degradation and reducing immune responses.
This formulation enables more effective systemic delivery of RNA-based therapeutics. It represents a key advancement in non-viral gene delivery systems. Many of these innovations are shaping the synthetic biology landscape. See our spotlight on Synthetic Biology Companies.
Similar Patents As EP2279254B1
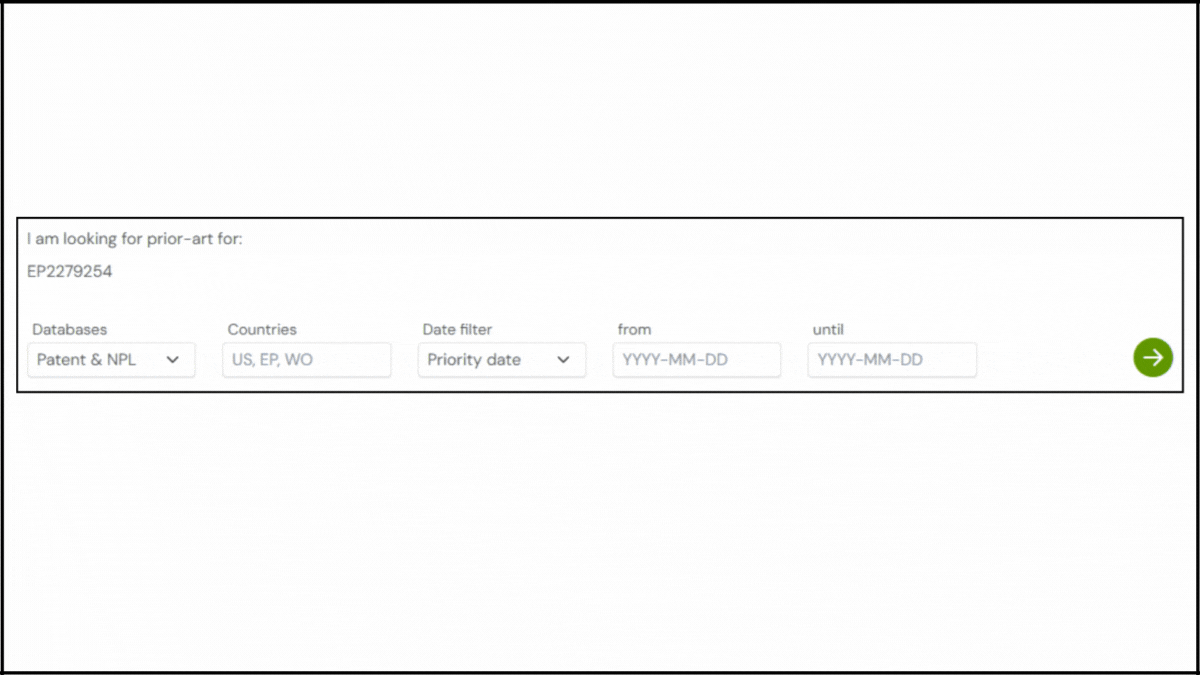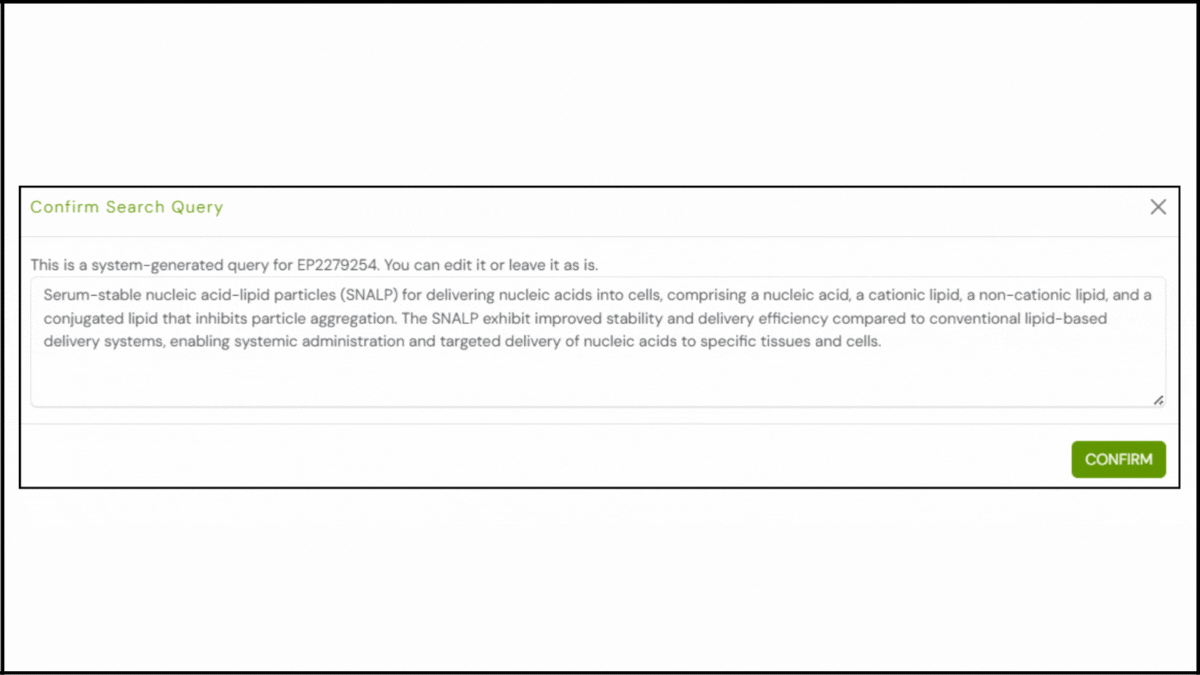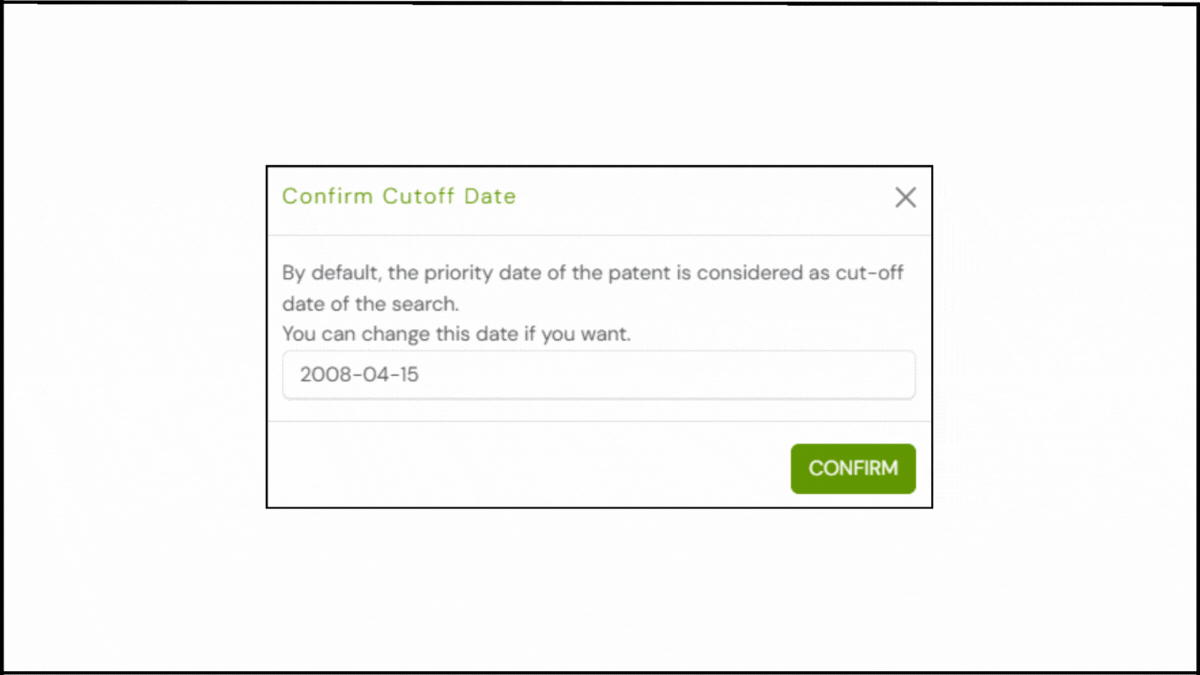
To explore the innovation landscape surrounding EP2279254B1, we ran the patent through the Global Patent Search tool. Below is a quick glimpse of the GPS tool in action:
Source: Global Patent Search
This analysis surfaced a list of related patents that share technical similarities in lipid composition, delivery efficiency, and serum stability. Below, we highlight four references that reflect comparable strategies in nucleic acid delivery. These examples provide insight into how similar challenges have been addressed across various systems.
#1. US2012021044A1
This US patent application, US2012021044A1, published in 2012, presents a novel cationic lipid composition designed to improve the delivery of nucleic acids such as siRNA into mammalian cells.
Developed by researchers at leading Korean institutions, the system emphasizes reduced cytotoxicity and enhanced intracellular uptake. It explores delivery via cationic liposomes, micelles, or emulsions, broadening the formulation possibilities for RNA therapeutics.

Source: GPS
What This Patent Introduces To The Landscape
- Distearoyl glutamide-based cationic lipids: Engineered lipids designed for reduced toxicity and improved delivery into animal cells.
- Use of DPhPE as a fusogenic phospholipid: Supports cell membrane interaction for efficient intracellular siRNA transport.
- Versatile formulation formats: Applicable as cationic liposomes, micelles, or emulsions for therapeutic nucleic acid delivery.
How It Connects To EP2279254B1
- Both patents focus on lipid-based delivery systems for nucleic acids, particularly siRNA.
- Each introduces a composition optimized for intracellular delivery with reduced toxicity.
- The emphasis on cationic lipid functionality and in vivo application reflects parallel innovation paths.
Why This Matters
This work showcases alternative strategies to enhance siRNA delivery while reducing toxicity. These goals align with the formulation objectives in EP2279254B1 and expand the innovation pathway in RNA therapeutics.
#2. US2011117125A1
This US patent application, US2011117125A1, published in 2011, presents a comprehensive platform for delivering nucleic acids using lipid-based carriers with controlled surface charge and optimized lipid ratios.
Developed by Tekmira, Alnylam, and the University of British Columbia, it focuses on delivering siRNA and other oligonucleotides effectively while maintaining serum stability and minimizing systemic toxicity.

Source: GPS
What This Patent Introduces To The Landscape
- Fully encapsulated lipid particles: Designed for enhanced serum stability and resistance to enzymatic degradation during systemic circulation.
- Optimized lipid molar ratios: Combines DLin-K-DMA, DSPC, cholesterol, and PEG-lipids to form neutral-surface nanoparticles.
- Adaptable targeting strategies: Includes antibody-conjugated liposomes and immunostimulatory payloads for potential vaccine delivery applications.
How It Connects To EP2279254B1
- Both patents prioritize serum-stable, non-toxic lipid nanoparticles for nucleic acid delivery.
- Each emphasizes defined lipid ratios and full nucleic acid encapsulation to boost therapeutic efficacy.
- The use of PEG-conjugated lipids to minimize aggregation and immune clearance is similar to the structural design in EP2279254B1.
Why This Matters
This patent reinforces key strategies in RNA therapeutic design, especially in creating balanced lipid compositions that ensure effective delivery, low toxicity, and robust in vivo stability.
#3. US6287591B1
This US patent, US6287591B1, published in 2001, discloses lipid-based formulations for encapsulating charged therapeutic agents, especially polyanionic compounds like nucleic acids.
Developed by Inex Pharmaceuticals, the system utilizes a four-component lipid mixture to improve drug-to-lipid ratios, enhance serum stability, and facilitate systemic delivery of a range of therapeutic agents.

Source: GPS
What This Patent Introduces To The Landscape
- Four-lipid delivery system: Combines amino lipids, neutral lipids, cholesterol, and PEG-lipids to encapsulate charged therapeutic agents.
- Neutral surface formulation: Designed to reduce rapid clearance and cytotoxicity associated with conventional cationic liposomes.
- High-efficiency nucleic acid encapsulation: Offers protection from degradation and supports repeat dosing in vivo.
How It Connects To EP2279254B1
- Both patents emphasize multi-lipid compositions to stabilize and deliver nucleic acids systemically.
- Each formulation includes PEG-conjugated lipids to reduce particle aggregation and prolong the circulation time.
- The approach to serum stability and encapsulation efficiency reflects shared design goals in non-viral RNA delivery.
Why This Matters
This early work laid the foundational principles for balancing lipid ratios to improve RNA encapsulation, biocompatibility, and circulation, key objectives echoed in EP2279254B1’s modern SNALP platform.
#4. US2001044147A1
This US patent application, US2001044147A1, published in 2001, describes stable polynucleotide delivery vehicles (SPDVs) comprising amphipathic cationic lipids complexed with non-cationic lipid components.
The invention focuses on enhancing DNA delivery to cells while reducing toxicity and preserving transfection efficiency after reconstitution or lyophilization, key attributes for therapeutic use.
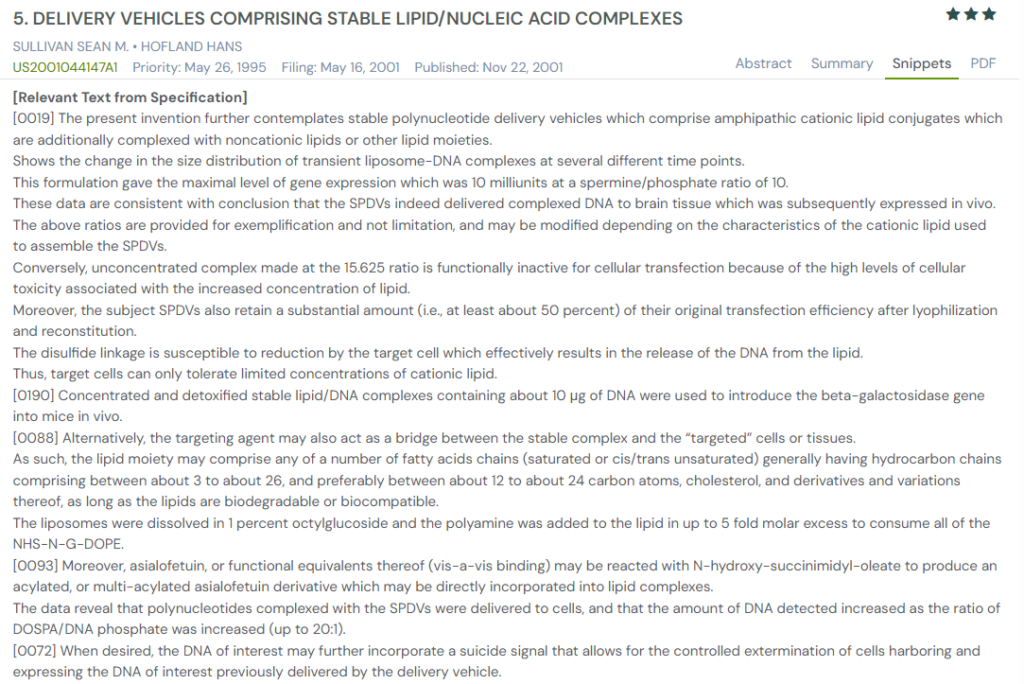
Source: GPS
What This Patent Introduces To The Landscape
- Amphipathic cationic lipid complexes: Designed to deliver DNA efficiently in vivo with reduced toxicity compared to conventional systems.
- Stability through lyophilization: SPDVs retain their transfection activity after freeze-drying and rehydration, a crucial property for storage and clinical use.
- Biodegradable lipid components: Uses biocompatible lipid chains to improve circulation and reduce immune response.
How It Connects To EP2279254B1
- Both focus on lipid-based delivery vehicles optimized for nucleic acid stability and intracellular delivery.
- Each leverages cationic and non-cationic lipid combinations to strike a balance between performance and safety.
- The emphasis on in vivo transfection and serum compatibility aligns with the therapeutic goals of EP2279254B1.
Why This Matters
This early patent showcases fundamental strategies in forming stable, rehydratable lipid-nucleic acid complexes, a concept that laid the groundwork for later advances in lipid nanoparticle delivery systems.
Recommended Read: To learn more about the frontrunners applying AI to therapeutic development, please read our blog 10 Leading Companies Using AI for Drug Discovery 2025.
How To Find Related Patents Using Global Patent Search
Understanding the broader patent landscape is key when evaluating nucleic acid delivery systems, lipid-based formulations, or non-viral gene therapy strategies. The Global Patent Search tool streamlines this process, helping users uncover technologies that share similar formulation goals or structural frameworks.
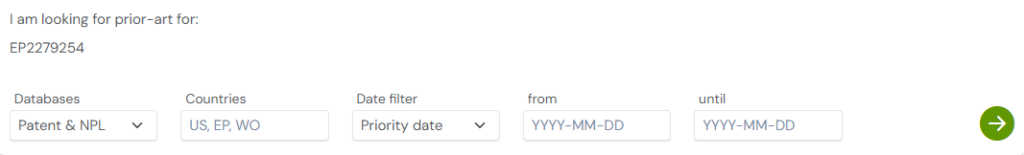
1. Enter the patent number into GPS: Start by entering a patent number like EP2279254B1 into the GPS tool. The platform transforms it into a smart query, which you can refine using terms like lipid composition, PEGylated particles, or siRNA delivery.
2. Explore conceptual snippets: Instead of comparing features claim-by-claim, GPS now offers curated text snippets. These highlight how other systems encapsulate nucleic acids, enhance serum stability, or reduce cytotoxicity.

3. Identify related inventions: The tool reveals patents that address similar formulation issues, like lipid ratios, encapsulation methods, or in vivo delivery, showing how others have tackled comparable challenges.
4. Compare systems, not legal claims: Rather than focusing on legal language, GPS compares real-world formulation strategies. This helps users trace scientific patterns without getting lost in claim structure.
5. Accelerate cross-domain insights: Whether you’re working in RNA therapeutics, drug delivery, or nanoparticle engineering, GPS lets you uncover related ideas across research domains that might otherwise remain disconnected.
AI-driven insights are also transforming how healthcare innovations like RNA delivery systems are developed. Explore more in our AI in Healthcare overview.
With this approach, Global Patent Search provides scientists, IP teams, and biotech developers with a concept-first method for exploring similar patents and making informed, data-focused decisions.
Disclaimer: The information provided in this article is for informational purposes only and should not be considered legal advice. The related patent references mentioned are preliminary results from the Global Patent Search tool and do not guarantee legal significance. For a comprehensive related patent analysis, we recommend conducting a detailed search using GPS or consulting a patent attorney.